When warmed above room temperature, Bromine will vaporise to form toxic red-brown bromine vapour, and iodine crystals will vaporise to form toxic purple iodine vapour.At room temperature, Iodineis a dark grey solid.Which option correctly describes the pattern of electronegativity and reactivity in metals Electronegativity decreases down a group, and reactivity. Which option correctly describes the pattern of valence electrons on the periodic table. At room temperature, Bromineis a dense red-brown liquid. simple chemical reactions online practice.Group 17 ( Halogens) are the most reactive nonmetals. At room temperature, Chlorineis a toxic, dense green gas. Metal elements are found on the left and in the middle of the periodic table. Reactivity also increases when moving from left to right in a row/period on the periodic table.At room temperature, Fluorineis a toxic yellow gas. 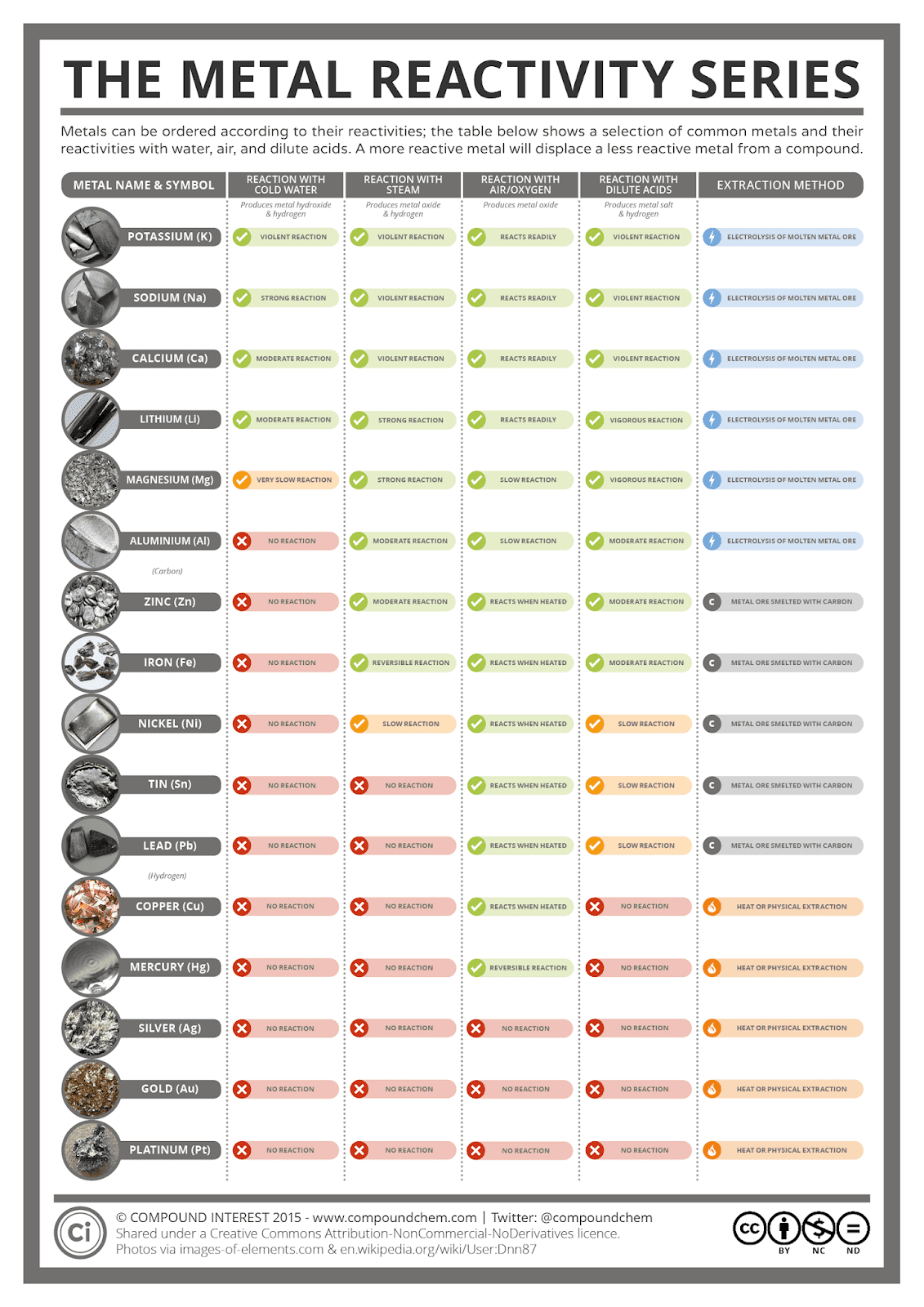
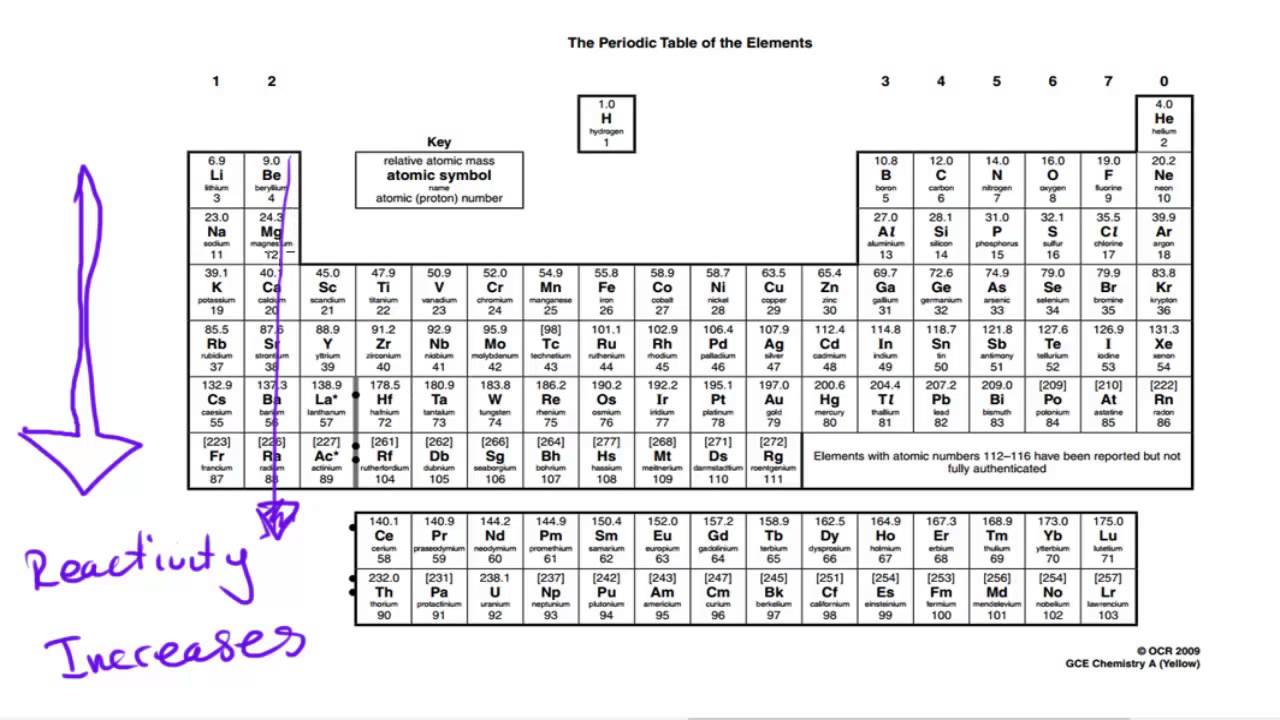
Down the group, the halogens show increasing relative atomic mass , and increasing melting and boiling points.There is also a trendin the physical properties of the halogen. By following the trend summary in the figure below, you. Within a compound, metal atoms have relatively low attraction for electrons, as indicated by their low electronegativities. Metals tend to lose electrons in chemical reactions, as indicated by their low ionization energies. They have a low density, and low melting and boiling points. Metallic character refers to the level of reactivity of a metal.Consequently: Metals are more reactive down the group as they can lose electrons more easily. Therefore, there is a weaker attraction between the nucleus and the electrons. Down the group, atoms get larger and their valence electrons are further away from the nucleus. He halogens also have similar physical properties: Explain reactivity in terms of electrons. (This is because the shells act as “shielding” from the positive charge of the nucleus) As the number of electron shells increases down group 7, it becomes harder to gain the last outer electron, and the reactivity of the elements decreases. The more electron shells there are between the outer electron and the nucleus, the harder it is to gain an additional outer electron. In chemical reactions, they gain one additional electron to gain a full outer shell. As they are in group 7, they each have seven electrons in their outer shell. This is due to the electronic structure of the halogens. This means that chlorine reacts less vigorously with sodium than fluorine, and bromine reacts less vigorously with sodium than chlorine. The chemical reactivity (how vigorously they react) of the halogens decreases down group 7. They react with water to produce metal hydroxide solutions (which are alkaline, hence the name of the group). Group 1 elements are also known as the alkali metals, and they have similar chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
